“Fueling the Future: Innovative Catalyst Solutions for the Oil, Gas and Petrochemical Industry’s Complex Challenges”
Hydrotreating Catalysts
Catalytic hydrotreating removes 90% of contaminants from liquid petroleum fractions to improve product quality and prevent equipment damage. The optimum catalyst depends on performance indicators, constraints, and reaction kinetics. Common catalyst metals include molybdenum and tungsten promoted by nickel and cobalt, while alumina is the most widely used support material. Hydrotreating catalysts can contain up to 26% active metal and 25% promotor components.

Products Catalyst
Isomerization Catalyst
Paraffin isomerization catalysts are employed to enhance the octane rating and diminish the benzene concentration of hydrocarbon streams that are utilized in the blending of gasoline. This chemical reaction transpires over a stationary bed of catalyst at moderate temperature and pressure.

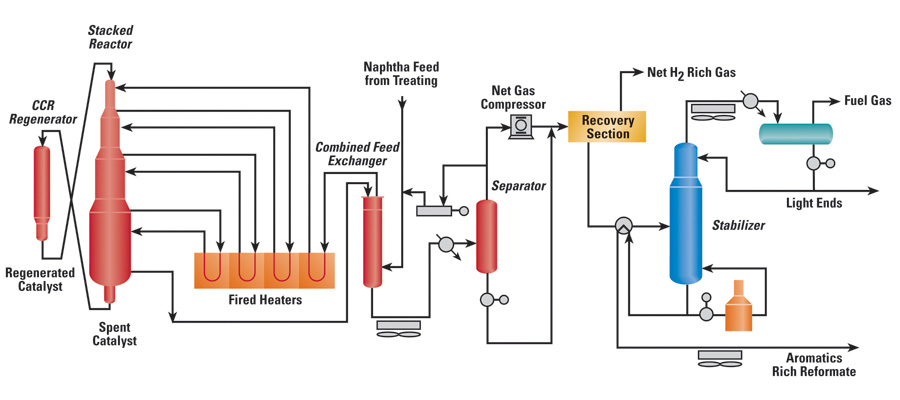
Reforming Catalyst type
Catalytic reforming is a chemical process that transforms low-octane petroleum refinery naphtha into high-octane reformates, which are premium blending stocks for high-octane gasoline. The process involves converting linear hydrocarbons into branched alkanes and cyclic naphthenes, which are then partially dehydrogenated to produce high-octane aromatic hydrocarbons. Reformate is not only a gasoline blending stock but also the primary source of bulk chemicals such as benzene, toluene, xylene, and ethylbenzene, which are used as raw materials for making plastics.
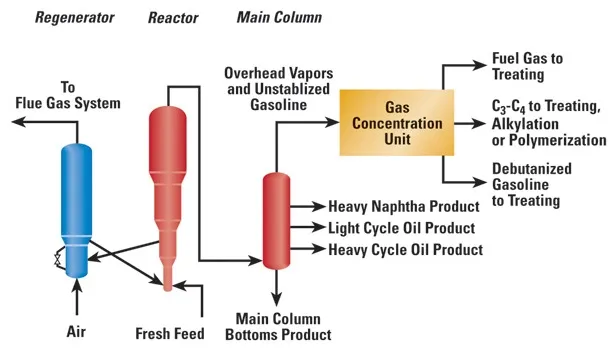
Fluid Catalytic Cracking (FCC)
Fluid Catalytic Cracking (FCC) is a refining process that converts heavy hydrocarbon molecules into lighter, more valuable products such as gasoline and diesel fuel. A modern FCC catalyst has four components: crystalline zeolite, matrix, binder, and filler.
Zeolite is the primary active component, which is a molecular sieve with a unique lattice structure that allows only certain size hydrocarbon molecules to enter. The desirable properties of an FCC catalyst include stability to high temperature and steam, high activity, large pore sizes, good resistance to attrition, and low coke production.
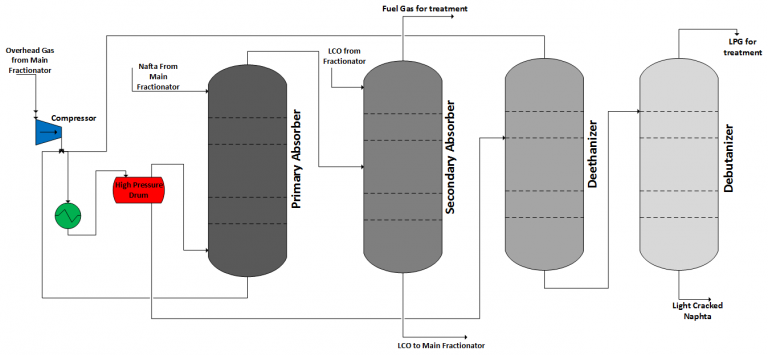
Residue Fluid Catalytic Cracking (RFCC)
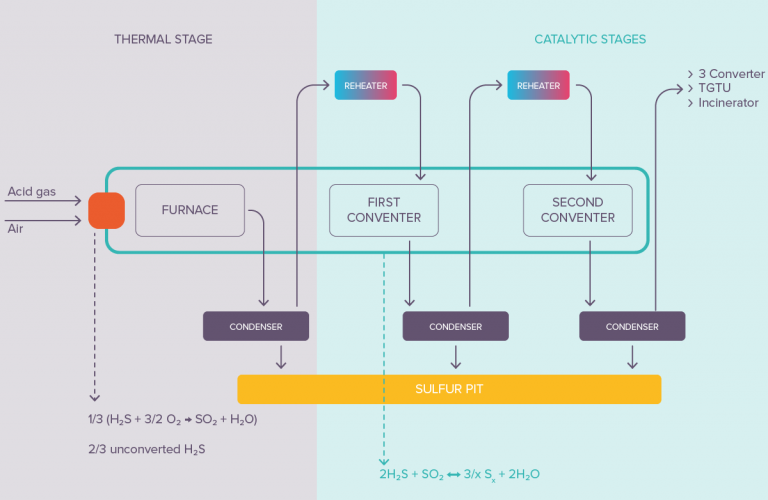
Sulphur Recovery (SRU)
Sulfur recovery units (SRUs) use “claus catalysts” to convert hydrogen sulfide (H2S) into elemental sulfur. Claus catalysts are typically made of alumina or silica, and are often promoted with metal oxides such as titanium oxide or zirconium oxide. The catalyst helps to speed up the chemical reactions that occur during the sulfur recovery process.
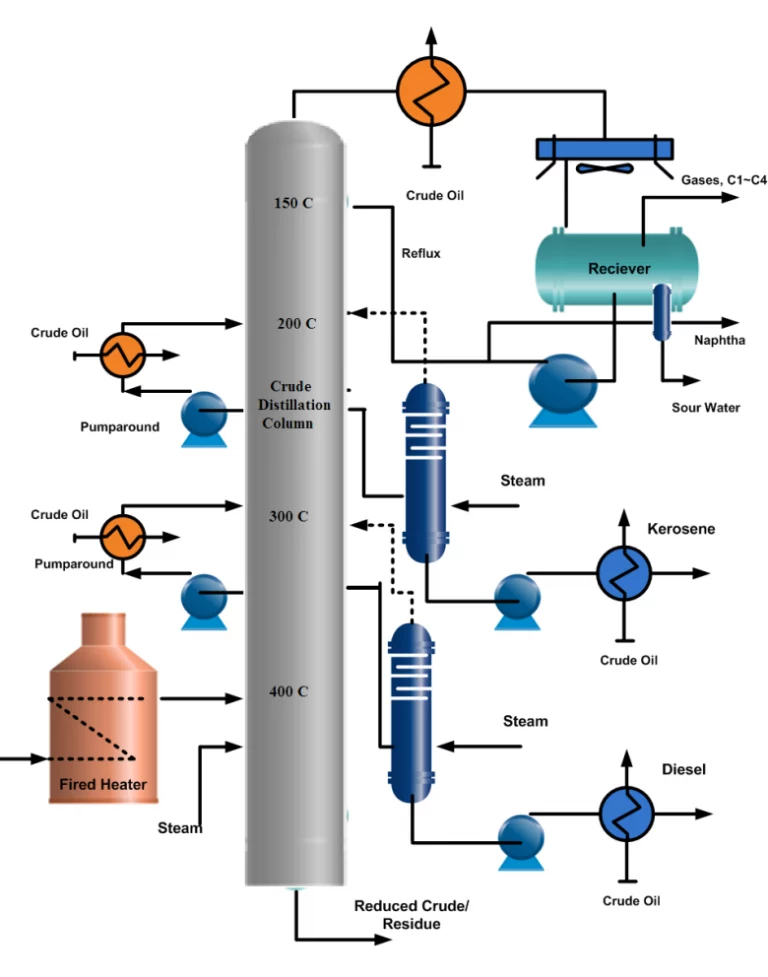
Reduced Crude Desulfurization (RCD)
Refiners are constantly seeking ways to convert heavy crude oil into lighter, more valuable products. One effective approach is to convert VGO hydrotreaters to VGO mild hydrocrackers. The RCD unifining process is another method that reduces impurities in heavy feedstocks, making them suitable for use as fuel oils or downstream processing. However, catalyst consumption has increased as feedstocks have become heavier. Nevertheless, improved catalysts and grading have helped to reduce the impact on operating factor. Accurate catalyst life prediction is crucial during the design phase of a new unit.
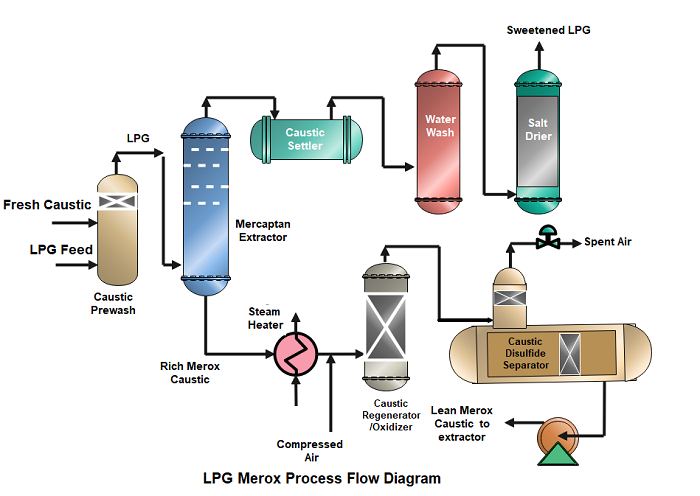
MEROX Catalyst
Merox is a trademarked name for a type of catalytic chemical process used in the petroleum refining industry to remove mercaptans from liquid hydrocarbons such as jet fuel, kerosene, and diesel fuel. The Merox process involves the use of an alkaline solution and a proprietary catalyst to oxidize mercaptans into disulfides, which can be easily removed from the fuel